Ruffell, S., Netzband, N., Bird, C., Young, A. H., & Juruena, M. F. (2020). The pharmacological interaction of compounds in ayahuasca: a systematic review. Brazilian Journal of Psychiatry, 42, 646-656. DOI:10.1590/1516-4446-2020-0884
Study Rationale
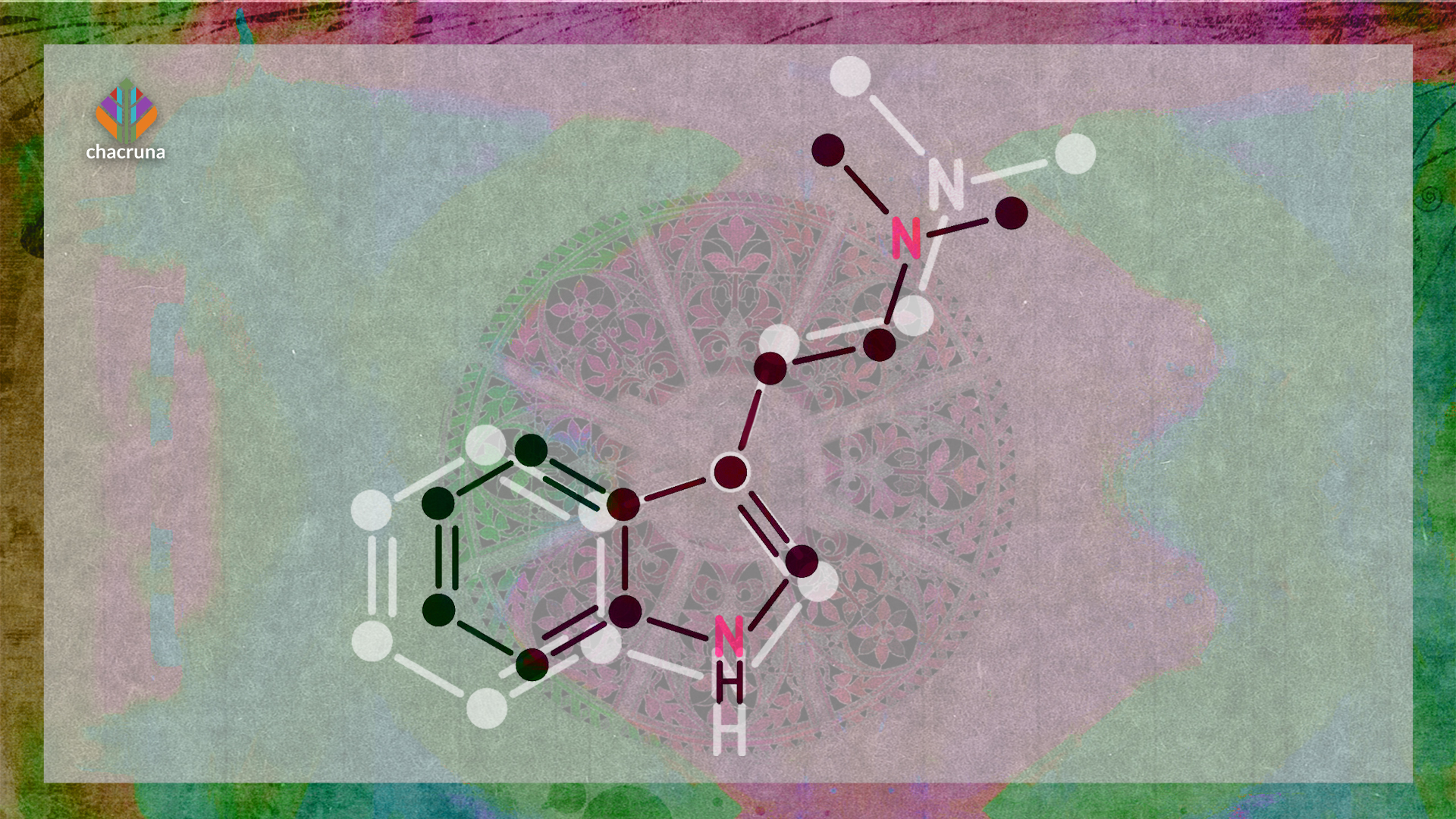
As various scholars and companies debate which chemicals in ayahuasca are required to achieve positive outcomes, questions remain regarding which components are essential, how they interact, and what happens if they are removed. The monoamine oxidase inhibitors (MAOIs) in ayahuasca allow for the bioavailability of N,N-Dimethyltryptamine (DMT) (Mckenna, 2004), however, it is currently unclear as to whether these compounds interact synergistically or merely additively. As such, a review of the available literature surrounding the pharmacology of ayahuasca was conducted.
Methodology
The PubMed, PsycINFO, and Web of Science databases were searched through September 2019 for the following terms: (ayahuasca OR DMT OR dimethyltryptamine) AND (B-carboline OR constituents OR chemistry OR harmine OR harmaline OR harmala alkaloids OR tetrahydroharmine OR harmalol OR MAOI OR monoamine oxidase inhibitor OR pharmacology OR pharmacokinetics OR pharmacodynamics OR psychopharmacology OR synergy). The reference lists of relevant studies were checked for additional papers, and secondary searches were performed using related keywords. A total of 2,141 papers were identified, of which 1,957 were extracted, following the removal of duplicates. A review of the titles and abstracts eliminated all but 202 papers, which were screened in greater detail for eligibility. The abstracts, methods, and findings of these papers were assessed, reducing the number to 57 for full text analysis. A total of 16 studies examined the pharmacology of ayahuasca as a brew or its known active compounds, either isolated or synergistically, and were included in the review. Only papers that had undergone full peer review and were published in English were included. The review followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines (Moher et al., 2009).
Summary of Results
Table 1
Summary of studies included in systematic review
| Authors | Title | Findings | Further comments |
| McKenna et al. (1984) | Monoamine oxidase inhibitors in South American hallucinogenic plants: tryptamine and ß-carboline constituents of ayahuasca | -Showed MAOI effect in vitro. -Proposes that inhibition experiments using mixtures of ß-carbolines indicate that their effects in combination are additive, rather than synergistic or antagonistic. | -Original proposal regarding DMT deamination prevention via MAOI in harmala alkaloids from P. viridis. |
| Callaway et al. (1994) | Platelet serotonin uptake sites increased in drinkers of ayahuasca | -Increased number of serotonin mRNA transporter sites in regular ayahuasca drinkers against control group. | -Shows an increased number of binding sites in platelets. -No evidence of this for DMT alone, which is suggestive of a synergistic effect. |
| Strassman et al. (1994) | Dose-response study of N, N-dimethyltryptamine in humans | -Peak DMT blood levels and subjective effects were seen within 2 minutes after drug administration and were negligible at 30 minutes. -DMT dose-dependently elevated blood pressure, heart rate, pupil diameter, and rectal temperature, in addition to elevating blood concentrations of ß-endorphin, corticotropin, cortisol, and prolactin. Growth hormone blood levels rose equally in response to all doses of DMT, and melatonin levels were unaffected. -Threshold doses for significant effects relative to placebo were also hallucinogenic (> 0.2 mg/kg) -Subjects exposed five or more times to 3,4-methylenedioxymethamphetamine had less robust pupil diameter effects than those exposed two times of less. -Evidence that DMT is unique in the inability to develop tolerance to its psychological effects. | -Dose-response data for IV DMT fumarate, neuroendocrine, cardiovascular, autonomic, and subjective effects in a group of experienced hallucinogen users. |
| Smith et al. (1998) | Agonist properties of N, N-dimethyltryptamine at serotonin 5-HT2A and 5-HT2C receptors | -DMT fully substituted for DOI. Intact choroid plexus was used to evaluate the agonist properties at endogenous 5-HT2C receptors. -DMT was a partial agonist at 5-HT2C receptors in this native preparation. -DMT behaves as an agonist at both 5-HT2A and 5-HT2C receptors. -One difference was evident in that the 5-HT2C, but not the 5-HT2A, receptor showed a profound desensitization to DMT over time (suggestive of limited application for repeat prescription). | -Evidence of DMT 5HT2a(c) agonism. |
| Callaway et al. (1999) | Pharmacokinetics of Hoasca alkaloids in healthy humans | -THH shows PK profile independent to harmine. -Affinities and other PK values provided. | -Evidence that THH alone may be a weak SSRI. -Implies further synergistic effects on the serotonin system. |
| Ott (1999) | Pharmahuasca: human pharmacology of oral DMT plus harmine | -MAO inhibition from simultaneous ingestion of ß-carbolines confirmed by eight self-experimenters. -Results of a total of some 70 bioassays are summarized and the literature on this subject is reviewed. | -Evidence that DMT and harmine in tablet form create similar effects to ayahuasca, further reinforcing the DMT MAOI interaction. -When orally ingested, DMT without harmine is non-active. |
| Glennon et al. (2000) | Binding of ß-carbolines and related agents at 5-HT2, 5-HT1A, dopamine (D2) and benzodiazepine receptors | -Affinity scores at 5-HT2 for harmine/harmaline. | -Shows that other harmala alkaloids also bind to the 5HT2 receptors, further suggesting synergistic potential in the serotonergic system. |
| Riba et al. (2003) | Human pharmacology of ayahuasca | -Diastolic blood pressure significant increase. -Heart rate moderate increase. -Increased urinary normetanephrine excretion. -Deaminated monoamine metabolite levels did-not decrease (contrary to typical MOAI effect profile). -The negligible harmine plasma levels found suggest a predominantly peripheral (gastrointestinal and liver) site of action for harmine. | -Double-blind placebo controlled clinical trial using freeze-dried ayahuasca. -PK angle. -Small sample size (n=18). |
| Riba et al. (2006) | Increased frontal and paralimbic activation following ayahuasca | -Significant activation of frontal and paralimbic brain regions. -Increased blood perfusion observed bilaterally in anterior insula, gather intensity in right hemisphere, and anterior cingulate/frontal medial cortex of right hemisphere. -Increases observed in left amygdala/parahippocampal gyrus. -Concludes that ayahuasca interacts with neural systems that are central to interoception and emotional processing. | -Double-blind placebo controlled clinical trial using freeze-dried ayahuasca. -Neuroimaging angle. -Used SPECT. |
| Fortunato et al. (2010) | Chronic administration of harmine elicits antidepressant-like effects and increases BDNF levels in rat hippocampus | -Increased BDNF protein levels in rat hippocampus. -Concludes that findings within support the hypothesis that harmine could bring about behavioural and molecular effects. | -Further evidence that the synergistic mechanisms of DMT + harmine are more than just effects of MAOIs. |
| Santos and Strassman (2011) | Autonomic, neuroendocrine, and immunological effects of ayahuasca: a comparative study with D-amphetamine | -Significant increases in prolactin. -Percentage of CD3/4 were decreased, natural killer cells increased. -Maximum changes occurred around 2 hours, returned to baseline after 24 hours. -Focuses on the synergistic effects of ayahuasca rather than individual action of compounds. -Immunological, rather than neuropsychological, perspective. | -Focuses on the synergistic effects of ayahuasca rather than individual action of compounds. -Immunological, rather than neuropsychological, perspective. |
| McIlhenny et al. (2011) | Methodology for determining the major constituents and metabolites of the Amazonian botanical medicine ayahuasca in human urine | -Showed that the major metabolite of a DMT is the corresponding DMT-NO, the first time this metabolite has been described in in vivo studies in humans. -Very little DMT detected in urine, despite the MAOI. -Major alkaloid excreted was THH. -List of other products and metabolites quantified. | -Provides methodology for identifying and quantifying constituents of ayahuasca in human urine. -PK data of tested samples provided. Excretion and metabolism of THH should be further investigated. |
| McIlhenny et al. (2012) | Methodology for determining major constituents of ayahuasca and its metabolites in blood | -DMT concentrations lower than DMT-NO at all time points. -Harmine and harmaline present in most samples. -Plasma DMT-NO concentrations three to four times higher than DMT. -DMT-NO forms rapidly after drug administration. | -Single methodology combining HPLC and gas chromatography to identify ayahuasca constituents in blood following oral consumption. -First report of presence of DMT-NO in human blood following ayahuasca/DMT administration. -Method for the most complete profile of DMT, harmala alkaloids, and metabolite concentrations. -THH levels peaked at around 4.5 hours. |
| Riba et al. (2012) | Metabolism and disposition of N, N-dimethyltryptamine and harmala alkaloids after oral administration of ayahuasca | -Less than 1% of DMT excreted unchanged. -The recovery of each harmala alkaloid plus its 0-demethylated metabolite varied greatly (between 9 and 65%). -Fifty per cent was recovered as indole-3-acetic acid or DMT-NO. -Ten per cent was other MAO-independent compounds. -Recovery of DMT plus metabolites reached 68%. -Harmol, harmalol, and THH conjugates were abundant in urine. | -PK study with implications regarding alternative metabolic routes for DMT other than biotransformation by MAO. -Freeze-dried ayahuasca. -Urine samples obtained. -Small sample (n=10). |
| Morales-García et al. (2017) | The alkaloids of Banisteriopsis caapi, the plant source of the Amazonian hallucinogen ayahuasca, stimulate adult neurogenesis in vitro | -Significant neurogenesis in adult hippocampal cells in vitro with harmine. | -Suggests that ayahuasca brew may have more complex synergistic properties than we currently understand. -Shows that harmine alone could be partially responsible for the neurological changes seen in ayahuasca users. |
| Sampedro et al. (2017) | Assessing the psychedelic “after-glow” in ayahuasca users: post-acute neurometabolic and functional connectivity changes are associated with enhanced mindfulness capacities | -Magnetic resonance spectroscopy showed post-acute reductions in glutamate + glutamine, creatine, and N-acetylaspartate+N- acetylaspartylglutamate in the posterior cingulate cortex. -Connectivity was increased between the posterior cingulate cortex and the anterior cingulate cortex, and between the anterior cingulate cortex and limbic structures in the right medial temporal lobe. -Glutamate + glutamine reductions correlated with increases in the “nonjudging” subscale of the Five Facets Mindfulness Questionnaire-Increased anterior cingulate cortex-medial temporal lobe connectivity correlated with increased scores on the self-compassion questionnaire. -Post-acute neural changes predicted sustained elevations in nonjudging 2 months later. | -DMN activity decrease and increased neural connectivity to other areas of the brain. -Supported by other studies on 5HT2a agonists. -Long-term neurological differences found after ayahuasca administration. |

Find more information on the upcoming Psychedelic Culture Conference.
Integration with extant literature
This systematic review shows that two of the constituents in ayahuasca, DMT and harmine, have been studied significantly more than the secondary harmala alkaloids (MAOIs). It appears there may be more synergistic mechanisms present than we currently understand. This is derived from data pertaining to individual constituents, which often shows overlapping biochemical and pharmacokinetic action. It is unclear at present whether these actions are of a true synergistic nature or are additive. Evidence suggests, despite the lack of solid data on synergy, that ayahuasca may have promising antidepressant qualities.
Increasing numbers of studies are being published focusing on DMT and other serotonin agonists (Carhart-Harris et al., 2021; Mitchell et al., 2021; Mithoefer et al., 2018; Palhano-Fontes et al., 2019; Ross et al., 2016; Rucker et al., 2019; Schartner & Timmermann, 2020; Strassman, 2000; Timmermann et al., 2019; Timmermann, Spriggs, et al., 2018). There is, however, a vast number of anthropological and qualitative articles which emphasise the importance of Banisteriopsis caapi. Traditional ayahuasca is composed of the Banisteriopsis caapi vine, with a DMT containing plant, usually Psychotria viridis, with the vine being the only consistent requirement for the brew (McKenna et al., 1984). In the Peruvian Amazon the term ayahuasca is also commonly used to indicate the vine only.
The harmala alkaloids reduce the deamination of DMT by MAO-A during first-pass metabolism, resulting in the subsequent absorption and distribution of this potent psychedelic (Yritia et al., 2002). The alkaloids are derived from β-carbolines, and their potential psychotropic properties have been discussed since research into psychedelic substances began (Naranjo, 1967). In recent times, researchers have started to focus their attention on the β-carbolines and their role in treating mental health conditions (Dos Santos & Hallak, 2017). The β-carbolines have been demonstrated to have a variety of biological actions, including anxiolytic, anticonvulsant and sedative effects, largely due to their interactions with serotonin and benzodiazepine receptors (Cao et al., 2007).
Papers that assess the levels of constituents in various brews and compare these to therapeutic outcomes have been summarised in Table 2. Due to the lack of heterogeneity between the study designs it is difficult to draw meaningful conclusions by comparing the outcomes of the studies. The table does however demonstrate that the ratio of constituents, as well as the dose of the brew, has an impact on mental health outcomes.
Table 2
Summary table of studies reporting constituency and psychometrics (i.e., depression, anxiety, wellbeing, and perceived peak experiences)
Study Design
Constituent
| Reference | Tradition | Design | Sample Size (n) | DMT (mg/ml) | THH (mg/ml) | HRL (mg/ml) | HRM (mg/ml) | Total Dosing Sessions |
| Osório et al. (2015) | Church – Santo Diame | Open label RCT | 6 | 0.8 | NA | NA | 0.21 | 1 |
| Gonzalez et al. (2021) | Traditional – Shipibo | Observational | 200 | 2 | 1-2 | 0.37-0.65 | 2 | 1 to 12 |
| González et al. (2020) | Traditional – Shipibo | Observational | 50 | 2 | 1-2 | 0.37-0.65 | 2 | 4 to 9 |
| Uthaug et al. (2018) | Neo-Shamanic | Observational | 30 | 1.90 | NA | 4.9 | 2.4 | 1 |
| Uthaug et al. (2018) | Shamanic | Observational | 27 | 0.95 | NA | 0.35 | 6.305 | 1 |
| Palhano-Fontes et al. (2019) | Church – Barquinha | Placebo Control RCT | 29 (14 active, 15 control) | 0.36 | 1.2 | 0.24 | 1.86 | 1 |
| Uthaug et al. (2021) | Neo-Shamanic | Placebo Control Observational | 30 (14 active, 16 control) | 3.6 | NA | 0.7 | 10.1 | 2 |
| Depression | P | Value | (Cohen’s D) | Anxiety: | P | Value | Wellbeing: | P | Value | Peak: | P Value | |||
| Reference | Scale | T1-T2 | T1-T3 | Dep T1-T4 | Scale | T1-T2 | T1-T3 | T1-T4 | Scale | T1-T2 | T1-T3 | T1-T4 | Scale | Peak T1 |
| Osório et al. (2015) | HAM-D MADRS | 0.01 0.01 | 0.01 0.003 | 0.11 0.009 | BPRS-AD | 0.02 | 0.02 | 0.02 | – | – | – | – | – | – |
| Gonzalez et al. (2021) | – | – | – | – | – | – | – | – | PWBS | 0.01 | 0.01 | 0.01 | – | – |
| González et al. (2020) | SA-45 D | 0.001 | 0.01 | 0.1 | SA-45 D | 0.001 | 0.001 | 0.01 | WHO-QOL-B | 0.001 | 0.001 | – | – | – |
| Uthaug et al. (2018) | DASS-21 | 0.001 | 0.001 | – | DASS-21 | NS | NS | – | SWLS | 0.027 | NS | – | EDI | 0.001 |
| Uthaug et al. (2018) | DASS-21 | 0.001 | 0.001 | – | DASS-21 | NS | NS | – | SWLS | 0.027 | NS | – | EDI | 0.001 |
| Palhano-Fontes et al. (2019) | MADRS HAM-D | 0.04 (0.84) | 0.04 (0.84) | 0.0001 (1.49) 0.019 (0.98) | – | – | – | – | – | – | – | – | MEQ HRS | 0.004 0.0001 |
| Uthaug et al. (2021) | DASS-21 | 0.017 | – | – | DASS-21 | 0.032 | – | – | – | – | – | – | EDI | NS |
It should be noted that some of the papers did not document tetrahydroharmine levels (Osório et al., 2015; Uthaug et al., 2021; Uthaug et al., 2018). This is the only constituent in the brew that has been demonstrated to function as an selective serotonin reuptake inhibitor (SSRI) and an MAOI, both used in the treatment of depression, with SSRIs being the current gold standard (Brunoni et al., 2009; Callaway et al., 1999). While it is unclear if this omission was from the brew altogether or simply from the constituency analysis, these particular studies coincidentally described non-significant changes in anxiety, wellbeing, and peak experience measures. Furthermore, due to inconsistencies in dose administration and consistency ratios between studies, the extent to which outcomes related to dose response and pharmacokinetics remains unclear. Nonetheless, these studies suggest that the harmala alkaloids do have a role in psychometric changes alongside DMT.

Shop our Collection of Psychedelic T-Shirts.
The Properties of the Harmala Alkaloids
Each of the harmala alkaloids has been associated with different effects, psychologically, pharmacologically, and pharmacokinetically. Table 3 summaries the therapeutic and psychoactive effects of each of the harmala alkaloids and Table 4 provides an overview of their pharmacokinetic profile.
Table 3
The therapeutic and psychoactive effects of the harmala alkaloids
| Therapeutic and psychoactive effects | Reference | Harmine | Harmaline | Tetrahydroharmine |
| Antidepressant properties | (McKenna et al., 1984) | X | X | X |
| Anxiolytic properties | (Ebrahimi-Ghiri et al., 2019) (Khan et al., 2013) | Not assessed | X | Not assessed |
| Increases serotonin and norepinephrine | (McKenna et al., 1984) | X | X | X |
| Raises dopamine levels in CNS | (Pimpinella & Palmery, 1995) | X | X | Not assessed |
| Anti-addictive properties | (Aricioglu-Kartal et al., 2003; Brierley & Davidson, 2012; Owaisat et al., 2012) | X | Not assessed | Not assessed |
| Diabetes management – mitogenic for human beta cells (Likely target DYRK1A) | (Wang et al., 2015) | X | Not assessed | Not assessed |
| Induce brain plasticity and neurogenesis | (Morales-García et al., 2017) | (Harmine metabolite harmol) | X | X |
| Upregulates 5-HT receptor density | (Callaway et al., 1994; McKenna et al., 1998) | Not assessed but unlikely (Graham et al., 1987) | Not assessed but unlikely (Graham et al., 1987) | X |
| Hallucinogenic properties (at high doses) | (Buckholtz & Boggan, 1977; Naranjo, 1967; River & Lindgren, 1972) | Gunn and Marshall (1920) suggest it is at 2mg/kg i.v. or 8mg/kg po | 1 mg./kg. i.v. or 4 mg./kg. po (Naranjo, 1967) | ?300mg but unclear (Gunn & Marshall, 1920) |
| Increases the number of nerve progenitor cells (may be important in treatment of damage by drug use) | (Dakic et al., 2016) | X | Not assessed | Not assessed |
Hallucinations, vomiting, confusion, and ataxia are thought to be due to central nervous system stimulation by MAOIs (Hamill et al., 2019). In a study by Glennon et al. (2000), the harmala alkaloids were found to bind to 5-HT2 receptors with a similar affinity to DMT. The psychedelic properties of both harmaline and harmine are thought to arise from their binding at the 5-HT receptors (Riba et al., 2003).
Table 4
| Overview of Pharmacokinetics | DMT | Harmine | Harmaline | Tetrahydroharmine |
| Pharmacokinetics profile (Callaway et al., 1999) | – | Pharmacokinetic profile correlates with that of DMT | – | Pharmacokinetic profile independent of harmine |
| Cmax (ng/ml) | 15.8 ± 4.4 | 114.8 ± 61.7 | 6.3 ± 3.1 | 91.0 ± 22.0 |
| Tmax (min) | 107.5 ± 32.5 | 102.0 ± 58.3 | 145.0 ± 66.9 | 174.0 ± 39.6 |
| AUC (mg min/ml) | 5.60 ± 4.53 | 22.88 ± 11.69 | – | 47.78 ± 25.88 |
| T 1/2 (min) | 259.4 ± 207.2 | 115.5 ± 60.1 | – | 531.9 ± 290.8 |
| IC50 (μM) (Passos et al., 2014) | – | MAO IC50 = 0.013 MAO-A IC50 = 0.002 MAO-B IC50 = 20 | MAO IC50 = 0.016 MAO-A IC50 = 0.003 MAO-B IC50 = 25 | MAO IC50 = 1.77 MAO-A IC50 = 0.074 MAO-B IC50 = 100 |
| 5HT2A binding capacity (Ki) (Glennon et al., 2000) | – | 397 | 5010 | >10000 |
Harmine
Of all the β-carboline alkaloids in ayahuasca, harmine is present in the highest concentration. It is associated with numerous therapeutic effects, such as astrocytic function restoration, anti-inflammatory effects, human neural progenitor cell the proliferation, and increases in levels of BDNF (Abelaira et al., 2013; Dos Santos & Hallak, 2017; Liu et al., 2017; Morales-Garcia et al., 2020; Morales-García et al., 2017). Other research has also indicated the role of harmine in the treatment of addiction, specifically in reducing relapse rates from methamphetamine, cocaine, and alcohol via dopamine neurotransmission (Aricioglu-Kartal et al., 2003; Owaisat et al., 2012). Harmine also displays affinity at both the DYRK1A and imidazoline I2 binding sites, a property which has potential in the pharmacological management of drug dependence (Brierley & Davidson, 2012). Dakic et al. (2016) also report that harmine exerts proliferative effects in human neural progenitors, by inhibiting DYRK1A. This has been suggested as a potential mechanism behind the antidepressant effects of the brew. Harmine may also have potential in the management of diabetes, and has shown potential in improving glycaemic control, increasing islet mass, and inducing beta cell proliferation (Wang et al., 2015).
Glutamine synthetase and glial-specific excitatory amino-acid transporter expression (glutamate transporter-1 (GLT-1), glutamate/aspartate transporter (GLAST)) can be altered in the brain tissue of those suffering from depression (Bernard et al., 2011; Choudary et al., 2005). Several different investigations utilising animal models have shown GLT-1 protein and gene expression are increased by harmine, as is the uptake of glutamate (Li et al., 2011; Liu et al., 2017; Sun et al., 2014). Harmine has also been found to protect against the effects of chronic unpredictable stress in mice, such as reduced levels of glial fibrillary acidic protein (Liu et al., 2017). This suggests that its antidepressant action may be due to the renewal of astrocytic function. In addition, Liu et al. (2017) found harmine enhanced outcomes in the forced swimming test and also protected mice against stress when undergoing the tail suspension test.
BDNF signal restoration is hypothesised to mediate the antidepressant effects of harmine. Fortunato and colleagues showed this by administering harmine to rats both acutely (Fortunato et al., 2009) and chronically (Fortunato et al., 2010), finding improvements in both the open field and forced swimming tests. Both studies also demonstrated increased hippocampal BDNF levels, when compared to treatment with imipramine. Liu et al. (2017) found when mice were exposed to chronic unpredictable stress and were not given harmine, levels of BDNF did not increase, nor did hippocampal neurogenesis occur. These findings suggest harmine may result in molecular and behavioural outcomes like antidepressants drugs.
Morales-García et al. (2017) found that tetrahydroharmine, harmaline, and harmol (the metabolite of harmine), induce neurogenesis in adults in vitro via neural stem cell differentiation, migration, and proliferation. The authors conclude that the Banisteriopsis caapi β-carbolines appear to have the ability to induce brain plasticity, thereby showing potential in the treatment of various neurological and psychiatric conditions. Interestingly, in a subsequent study Morales-Garcia et al. (2020) showed that DMT led to the generation of new neurones via activation of the sub granular hippocampal dentate gyrus – the predominant neurogenic niche in adults. This research, conducted in mice, showed subsequent improvements in memory, demonstrating the functional relevance of the above findings. Furthermore, SIGMAR-1 activation appeared to underlie the neurogenic effects of DMT. The authors conclude that DMT induces the growth of new hippocampal neurones, promotes the migration of neuroblasts and regulates neural stem cell proliferation, largely via sub granular neurogenic niche activation, improving memory and spatial learning tasks as well as inducing neurogenesis (Morales-Garcia et al., 2020). The authors further highlight the potential antidepressant properties associated with neurogenesis, with DMT showing a considerably more potent neurogenic profile than the β-carbolines (Morales-Garcia et al., 2020; Morales-García et al., 2017).
Harmaline
Harmaline has been demonstrated to have numerous pharmacological functions including hypothermic and vasorelaxant activity, antitumoral, antimicrobial, antiplatelet, antileishmanial, and antiplasmodial effects (Khan et al., 2013). It has been found to be effective in managing various conditions associated with microbes, such as Candida albicans, Proteus vulgaris, Staphylococcus aureus, Aspergillus niger, and Escherichia coli (Wink et al., 1998). Harmaline has been shown to minimise the proliferation of cells in vitro when investigating promyelocytic cell lines in humans, with the optimal dose being 6–10 µg/mL and higher doses of 15–30 µg/mL generally considered cytotoxic (Zaker et al., 2007). Naranjo (1967) found harmaline was hallucinogenic at 4 mg/kg when taken orally – about half the amount required to achieve psychedelic effects when compared to harmine. Of all the harmala alkaloids, harmaline is usually present in the lowest concentrations. Despite this, it exerts various pharmacological effects, such as antimicrobial and vasorelaxant properties, as well as being an anxiolytic and an antidepressant (Ebrahimi-Ghiri et al., 2019; Khan et al., 2013).
Tetrahydroharmine
Platelet serotonin uptake sites appear to increase in those who drink ayahuasca. This is thought to be associated with positive mental health effects, largely as this is deemed to be indicative of neuronal serotonin uptake activity, although the degree to which this actually reflects neuronal activity is debated (Callaway et al., 1994). Callaway hypothesised that tetrahydroharmine was responsible for the upregulation of 5HT uptake sites. He therefore commenced daily dosing with tetrahydroharmine over a six-week period, conducting SPECT scans of his own brain before and after the period of treatment. Central 5-HT receptor density was found to increase in his prefrontal cortex. Upon ceasing to consume tetrahydroharmine, SPECT scans revealed over several weeks that the density slowly returned to pre-dosing levels. Despite clear methodological issues, this one-man experiment suggests tetrahydroharmine may have potentially significant effects (McKenna et al., 1998).
Interestingly, tetrahydroharmine is the only component of the brew that is known to function as an SSRI, albeit weakly (Callaway et al., 1999). The psychoactive effects induced by tetrahydroharmine are less prominent than that of harmine, with harmaline having the strongest effect of all the harmala alkaloids (Naranjo, 1967). Gunn and Marshall (1920) provided an oral administration of 300mg tetrahydroharmine to a single volunteer, who described similar hallucinogenic effects to 100mg of harmaline. Although limited conclusions can be derived from this single experiment, the results suggest the hallucinogenic effects of tetrahydroharmine are roughly one-third that of harmaline.
Callaway et al. (1999) noted that in the ayahuasca-using church, the UDV, teas with higher levels of tetrahydroharmine relative to harmine and harmaline were favoured by both church elders and the congregation.Findings in the Santo Daime confirm this observation (Kaasik et al., 2021). Callaway connected the variation of the relative concentration of tetrahydroharmine in ayahuasca brews to its variability in Banisteriopsis caapi. Kaasik et al. (2021) show that it may also depend on the preparation of the brew. Tetrahydroharmine is the reduction product of harmaline, and consequently levels of THH are generally found to be higher in preparations that are brewed for longer (Kaasik et al., 2021).
Although no quantitative data exists comparing the therapeutic effects of DMT combined with other harmala alkaloid containing plants such as Peganum harmala (Syrian Rue) to Banisteriopsis caapi, reports describing subjective experience can be consulted. Differences appear to exist and have been recorded in various forums, such as online forums and psychonautical guides to plant medicine: ‘substituting, say, Syrian rue for the ayahuasca vine, even though the rue contains the same harmala alkaloids, does apparently make an experiential difference. The experience with rue has been described as crystalline, cold, overwhelming, erratic, and uncaring, compared with that of the ayahuasca vine, which has been described as warm, organic, friendly, and purposeful’ (Beyer, 2009)p253. It should however be noted that the ratios in Syrian Rue are quite different to that of Banisteriopsis caapi, with Rue containing much higher levels of harmaline and lower levels of tetrahydroharmine (Moloudizargari et al., 2013). It is possible that this difference in ratio accounts for the qualitatively different experiences suggested above.
Study Relevance
Prior to this study there had not been a systematic attempt to elucidate the pharmacological basis of ayahuasca. Not only is this necessary to better understand its therapeutic effects and safety profile, but also to determine how specific components in the brew result in therapeutic outcomes.

Discover the Indigenous Reciprocity Initiative of the Americas
References
Abelaira, H. M., Réus, G. Z., & Quevedo, J. (2013). Animal models as tools to study the pathophysiology of depression. Brazilian Journal of Psychiatry, 35, S112-S120.
Aricioglu-Kartal, F., Kayır, H., & Uzbay, I. T. (2003). Effects of harman and harmine on naloxone-precipitated withdrawal syndrome in morphine-dependent rats. Life sciences, 73(18), 2363-2371.
Bernard, R., Kerman, I. A., Thompson, R. C., Jones, E. G., Bunney, W. E., Barchas, J. D., Schatzberg, A. F., Myers, R. M., Akil, H., & Watson, S. J. (2011). Altered expression of glutamate signaling, growth factor, and glia genes in the locus coeruleus of patients with major depression. Molecular psychiatry, 16(6), 634-646.
Beyer, S., V. (2009). Singing to the Plants: A Guide to Mestizo Shamanism in the Upper Amazon. University of New Mexico Press.
Brierley, D. I., & Davidson, C. (2012). Developments in harmine pharmacology—Implications for ayahuasca use and drug-dependence treatment. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 39(2), 263-272.
Brunoni, A. R., Fraguas Jr, R., & Fregni, F. (2009). Pharmacological and combined interventions for the acute depressive episode: focus on efficacy and tolerability. Therapeutics and clinical risk management, 5, 897.
Buckholtz, N. S., & Boggan, W. O. (1977). Monoamine oxidase inhibition in brain and liver produced by β-carbolines: structure-activity relationships and substrate specificity. Biochemical Pharmacology, 26(21), 1991-1996.
Callaway, J. C., Airaksinen, M. M., McKenna, D. J., Brito, G. S., & Grob, C. S. (1994). Platelet serotonin uptake sites increased in drinkers of ayahuasca. Psychopharmacology, 116(3), 385-387.
Callaway, J. C., McKenna, D. J., Grob, C. S., Brito, G. S., Raymon, L., Poland, R. E., Andrade, E., Andrade, E., & Mash, D. C. (1999). Pharmacokinetics of Hoasca alkaloids in healthy humans. Journal of Ethnopharmacology, 65(3), 243-256.
Cao, R., Peng, W., Wang, Z., & Xu, A. (2007). β-Carboline alkaloids: biochemical and pharmacological functions. Current medicinal chemistry, 14(4), 479-500.
Choudary, P. V., Molnar, M., Evans, S., Tomita, H., Li, J., Vawter, M., Myers, R., Bunney, W., Akil, H., & Watson, S. (2005). Altered cortical glutamatergic and GABAergic signal transmission with glial involvement in depression. Proceedings of the National Academy of Sciences, 102(43), 15653-15658.
Dakic, V., de Moraes Maciel, R., Drummond, H., Nascimento, J. M., Trindade, P., & Rehen, S. K. (2016). Harmine stimulates proliferation of human neural progenitors. PeerJ, 4, e2727.
Dos Santos, R. G., & Hallak, J. E. (2017). Effects of the natural β-carboline alkaloid harmine, a main constituent of ayahuasca, in memory and in the hippocampus: A systematic literature review of preclinical studies. Journal of psychoactive drugs, 49(1), 1-10.
dos Santos, R. G., & Strassman, R. (2011). Ayahuasca and psychosis. The ethnopharmacology of ayahuasca. Kerala: Transworld Research Network, 97-99.
Ebrahimi-Ghiri, M., Nasehi, M., & Zarrindast, M.-R. (2019). Anxiolytic and antidepressant effects of ACPA and harmaline co-treatment. Behavioural Brain Research, 364, 296-302. https://doi.org/https://doi.org/10.1016/j.bbr.2019.02.034
Fortunato, J. J., Réus, G. Z., Kirsch, T. R., Stringari, R. B., Fries, G. R., Kapczinski, F., Hallak, J. E., Zuardi, A. W., Crippa, J. A., & Quevedo, J. (2010). Chronic administration of harmine elicits antidepressant-like effects and increases BDNF levels in rat hippocampus. Journal of neural transmission, 117(10), 1131-1137.
Fortunato, J. J., Réus, G. Z., Kirsch, T. R., Stringari, R. B., Stertz, L., Kapczinski, F., Pinto, J. P., Hallak, J. E., Zuardi, A. W., & Crippa, J. A. (2009). Acute harmine administration induces antidepressive-like effects and increases BDNF levels in the rat hippocampus. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 33(8), 1425-1430.
Glennon, R. A., Dukat, M., Grella, B., Hong, S.-S., Costantino, L., Teitler, M., Smith, C., Egan, C., Davis, K., & Mattson, M. V. (2000). Binding of β-carbolines and related agents at serotonin (5-HT2 and 5-HT1A), dopamine (D2) and benzodiazepine receptors. Drug and Alcohol Dependence, 60(2), 121-132.
Gonzalez, D., Cantillo, J., Perez, I., Carvalho, M., Aronovich, A., Farre, M., Feilding, A., Obiols, J. E., & Bouso, J. C. (2021). The shipibo ceremonial use of ayahuasca to promote well-being: an observational study. Frontiers in Pharmacology, 12, 1059.
González, D., Cantillo, J., Pérez, I., Farré, M., Feilding, A., Obiols, J. E., & Bouso, J. C. (2020). Therapeutic potential of ayahuasca in grief: a prospective, observational study. Psychopharmacology (Berl), 1-12.
Graham, D., Tahraoui, L., & Langer, S. (1987). Effect of chronic treatment with selective monoamine oxidase inhibitors and specific 5-hydroxytryptamine uptake inhibitors on [3H] paroxetine binding to cerebral cortical membranes of the rat. Neuropharmacology, 26(8), 1087-1092.
Gunn, J., & Marshall, D. (1920). The Harmala Alkaloids in Malaria. Proc. Roy. Soc. of Edinburgh,
Hamill, J., Hallak, J., Dursun, S. M., & Baker, G. (2019). Ayahuasca: psychological and physiologic effects, pharmacology and potential uses in addiction and mental illness. Current neuropharmacology, 17(2), 108-128.
Kaasik, H., Souza, R. C., Zandonadi, F. S., Tófoli, L. F., & Sussulini, A. (2021). Chemical composition of traditional and analog ayahuasca. Journal of Psychoactive Drugs, 53(1), 65-75.
Khan, F. A., Maalik, A., Iqbal, Z., & Malik, I. (2013). Recent pharmacological developments in β-carboline alkaloid “harmaline”. European journal of pharmacology, 721(1-3), 391-394.
Li, Y., Sattler, R., Yang, E. J., Nunes, A., Ayukawa, Y., Akhtar, S., Ji, G., Zhang, P.-W., & Rothstein, J. D. (2011). Harmine, a natural beta-carboline alkaloid, upregulates astroglial glutamate transporter expression. Neuropharmacology, 60(7-8), 1168-1175.
Liu, F., Wu, J., Gong, Y., Wang, P., Zhu, L., Tong, L., Chen, X., Ling, Y., & Huang, C. (2017). Harmine produces antidepressant-like effects via restoration of astrocytic functions. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 79, 258-267.
McIlhenny, E. H., Riba, J., Barbanoj, M. J., Strassman, R., & Barker, S. A. (2011). Methodology for and the determination of the major constituents and metabolites of the Amazonian botanical medicine ayahuasca in human urine. Biomedical Chromatography, 25(9), 970-984.
McIlhenny, E. H., Riba, J., Barbanoj, M. J., Strassman, R., & Barker, S. A. (2012). Methodology for determining major constituents of ayahuasca and their metabolites in blood. Biomedical Chromatography, 26(3), 301-313.
McKenna, D. J., Callaway, J. C., & Grob, C. S. (1998). The scientific investigation of Ayahuasca: a review of past and current research. The Heffter Review of Psychedelic Research, 1(65-77), 195-223.
McKenna, D. J., Towers, G. N., & Abbott, F. (1984). Monoamine oxidase inhibitors in South American hallucinogenic plants: tryptamine and β-carboline constituents of ayahuasca. Journal of Ethnopharmacology, 10(2), 195-223.
Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G., & Group*, P. (2009). Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Annals of internal medicine, 151(4), 264-269.
Moloudizargari, M., Mikaili, P., Aghajanshakeri, S., Asghari, M. H., & Shayegh, J. (2013). Pharmacological and therapeutic effects of Peganum harmala and its main alkaloids. Pharmacognosy reviews, 7(14), 199. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3841998/pdf/PRev-7-199.pdf
Morales-Garcia, J. A., Calleja-Conde, J., Lopez-Moreno, J. A., Alonso-Gil, S., Sanz-SanCristobal, M., Riba, J., & Perez-Castillo, A. (2020). N, N-dimethyltryptamine compound found in the hallucinogenic tea ayahuasca, regulates adult neurogenesis in vitro and in vivo. Translational psychiatry, 10(1), 1-14.
Morales-García, J. A., de la Fuente Revenga, M., Alonso-Gil, S., Rodríguez-Franco, M. I., Feilding, A., Perez-Castillo, A., & Riba, J. (2017). The alkaloids of Banisteriopsis caapi, the plant source of the Amazonian hallucinogen Ayahuasca, stimulate adult neurogenesis in vitro. Sci Rep, 7(1), 1-13.
Naranjo, C. (1967). Ayahuasca, caapi, yage. Psychotropic properties of the harmala alkaloids. Psychopharmacology bulletin, 4(3), 16-17.
Osório, F. d. L., Sanches, R. F., Macedo, L. R., Dos Santos, R. G., Maia-de-Oliveira, J. P., Wichert-Ana, L., De Araujo, D. B., Riba, J., Crippa, J. A., & Hallak, J. E. (2015). Antidepressant effects of a single dose of ayahuasca in patients with recurrent depression: a preliminary report. Brazilian Journal of Psychiatry, 37, 13-20.
Ott, J. (1999). Pharmahuasca: human pharmacology of oral DMT plus harmine. Journal of Psychoactive Drugs, 31(2), 171-177.
Owaisat, S., Raffa, R. B., & Rawls, S. M. (2012). In vivo comparison of harmine efficacy against psychostimulants: preferential inhibition of the cocaine response through a glutamatergic mechanism. Neuroscience letters, 525(1), 12-16.
Palhano-Fontes, F., Barreto, D., Onias, H., Andrade, K. C., Novaes, M. M., Pessoa, J. A., Mota-Rolim, S. A., Osório, F. L., Sanches, R., & Dos Santos, R. G. (2019). Rapid antidepressant effects of the psychedelic ayahuasca in treatment-resistant depression: a randomized placebo-controlled trial. Psychological medicine, 49(4), 655-663.
Passos, C. D. S., Simoes-Pires, C., Henriques, A., Cuendet, M., Carrupt, P.-A., & Christen, P. (2014). Alkaloids as inhibitors of monoamine oxidases and their role in the central nervous system. In Studies in Natural Products Chemistry (Vol. 43, pp. 123-144). Elsevier.
Pimpinella, G., & Palmery, M. (1995). Interaction of β-carbolines with central dopaminergic transmission in mice: structure-activity relationships. Neuroscience letters, 189(2), 121-124.
Riba, J., McIlhenny, E. H., Valle, M., Bouso, J. C., & Barker, S. A. (2012). Metabolism and disposition of N, N‐dimethyltryptamine and harmala alkaloids after oral administration of ayahuasca. Drug Testing and Analysis, 4(7-8), 610-616.
Riba, J., Romero, S., Grasa, E., Mena, E., Carrió, I., & Barbanoj, M. J. (2006). Increased frontal and paralimbic activation following ayahuasca, the pan-Amazonian inebriant. Psychopharmacology, 186(1), 93-98.
Riba, J., Valle, M., Urbano, G., Yritia, M., Morte, A., & Barbanoj, M. J. (2003). Human pharmacology of ayahuasca: subjective and cardiovascular effects, monoamine metabolite excretion, and pharmacokinetics. Journal of Pharmacology and Experimental Therapeutics, 306(1), 73-83.
River, L., & Lindgren, J. (1972). Ayahuasca, The South American Hallucinigenic drink: ethnobotanical and chemical investigations. Economic Botany, 29, 101-129.
Sampedro, F., de la Fuente Revenga, M., Valle, M., Roberto, N., Domínguez-Clavé, E., Elices, M., Luna, L. E., Crippa, J. A. S., Hallak, J. E., & De Araujo, D. B. (2017). Assessing the psychedelic “after-glow” in ayahuasca users: post-acute neurometabolic and functional connectivity changes are associated with enhanced mindfulness capacities. International Journal of Neuropsychopharmacology, 20(9), 698-711.
Smith, R. L., Canton, H., Barrett, R. J., & Sanders-Bush, E. (1998). Agonist properties of N, N-dimethyltryptamine at serotonin 5-HT2A and 5-HT2C receptors. Pharmacology Biochemistry and Behavior, 61(3), 323-330.
Strassman, R. J., Qualls, C. R., Uhlenhuth, E. H., & Kellner, R. (1994). Dose-response study of N, N-dimethyltryptamine in humans: II. Subjective effects and preliminary results of a new rating scale. Archives of general psychiatry, 51(2), 98-108.
Sun, P., Zhang, S., Li, Y., & Wang, L. (2014). Harmine mediated neuroprotection via evaluation of glutamate transporter 1 in a rat model of global cerebral ischemia. Neuroscience letters, 583, 32-36.
Uthaug, M., Mason, N., Toennes, S., Reckweg, J., Perna, E. d. S. F., Kuypers, K., van Oorsouw, K., Riba, J., & Ramaekers, J. (2021). A placebo-controlled study of the effects of ayahuasca, set and setting on mental health of participants in ayahuasca group retreats. Psychopharmacology, 1-12.
Uthaug, M., Van Oorsouw, K., Kuypers, K., Van Boxtel, M., Broers, N., Mason, N., Toennes, S., Riba, J., & Ramaekers, J. (2018). Sub-acute and long-term effects of ayahuasca on affect and cognitive thinking style and their association with ego dissolution. Psychopharmacology (Berl), 235(10), 2979-2989. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6182612/pdf/213_2018_Article_4988.pdf
Wang, P., Alvarez-Perez, J.-C., Felsenfeld, D. P., Liu, H., Sivendran, S., Bender, A., Kumar, A., Sanchez, R., Scott, D. K., & Garcia-Ocaña, A. (2015). A high-throughput chemical screen reveals that harmine-mediated inhibition of DYRK1A increases human pancreatic beta cell replication. Nature Medicine, 21(4), 383-388.
Wink, M., Schmeller, T., & Latz-Brüning, B. (1998). Modes of action of allelochemical alkaloids: interaction with neuroreceptors, DNA, and other molecular targets. Journal of chemical Ecology, 24(11), 1881-1937.
Yritia, M., Riba, J., Ortuno, J., Ramirez, A., Castillo, A., Alfaro, Y., de la Torre, R., & Barbanoj, M. J. (2002). Determination of N, N-dimethyltryptamine and β-carboline alkaloids in human plasma following oral administration of ayahuasca. Journal of Chromatography B, 779(2), 271-281.
Zaker, F., Oody, A., & Arjmand, A. (2007). A study on the antitumoral and differentiation effects of Peganum harmala derivatives in combination with ATRA on leukaemic cells. Archives of pharmacal research, 30(7), 844-849.
Art by Mariom Luna.